Medical Imaging: Sonography
Medical imaging is understood to mean instrumental diagnostic procedures for the visual representation of a clinical finding.
These procedures include X-ray, sonography (ultrasound), angiography (radiological imaging of blood and lymph vessels), CT (computer tomography), MRI (magnetic resonance imaging), PET (positron emission tomography) etc. etc.
In some cases, individual methods are combined (e. g. PET-CT) to obtain a more meaningful result. The image data is either two- or three-dimensional, depending on the procedure, and provides a representation of organs and structures of the body in order to be able to precisely identify disease-related changes.
Imaging is not only necessary for the detection of disease-related changes, but also for monitoring the progression of a disease or the success of a therapy. In addition, imaging aids safe punctures, tissue ablation and minimally invasive procedures.
Imaging is also indispensable to prepare and plan an exact irradiation in order to damage as little healthy tissue as possible. A better image presentation can be achieved during imaging, if contrast agents are used. Imaging, which makes structures and processes in the human body visible, has therefore long since become an indispensable part of medicine.
The first part deals with sonography, which is most commonly used in imaging.
This procedure has the decisive advantage that there is no radiation exposure whatsoever, so that it can be repeated as often as required without stressing the body.
The ultrasound probe, which is placed on the skin from the outside, works with sound waves that are outwith human hearing.
These ultrasound waves hit the tissue and are scattered, refracted and reflected.
A gel applied to the skin (viscous contact gel) ensures that the ultrasonic waves emitted by the transducer are transmitted to the tissue.
The transducer is both transmitter and receiver of ultrasonic waves. Sonography works according to the echo-principle, whereby the ultrasound emitted is reflected differently by the layers of the examined object.
Different transducers
The device calculates the layer structure of the examined object from the echo time of the reflected signals. There are diverse transducers, which differ not only in their shape but also in the range of frequency, which results in a lower or deeper penetration depth for the sound waves. Deep penetrating frequencies are used for the abdominal area. The strength of the respective reflection of the sound waves (echogenicity) is then visually represented by different white-gray-black shades on the monitor.
Not everything can be well depicted on ultrasoound
All liquid and soft structures such as thyroid, testicles, heart, liver, gall bladder, kidneys etc. can be well represented. In contrast, all structures containing air or surrounded by hard tissue, such as lungs, trachea, intestines, etc., can be poorly represented.
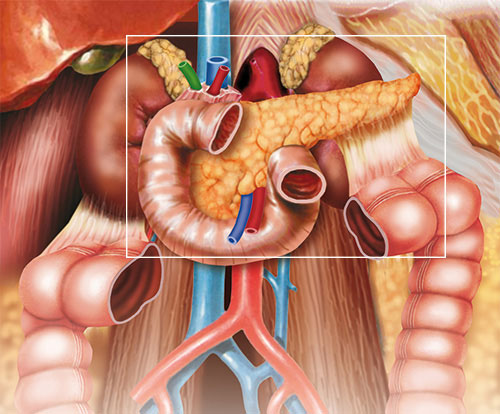
Since the pancreas lies in the back of the abdomen and is often covered by parts of the stomach and intestines that contain air or rather flatulence, it is sometimes difficult to image. The overlapping air obstructs an accurate image.
In certain cases, the accuracy of the sonography can be enhanced by a contrasting agent (CEUS).
Another possibility is colour Doppler sonography, which is used for vascular examination. This method works with the physical Doppler Effect (e.g. approaching and disappearing siren) and thus enables an exact localisation of circulatory disorders. It is also possible to visualize small blood vessels that are not visible using conventional methods.
An especially effective examination method is endosonography, in which the transducer is not placed on the skin from the outside but inserted into the body.
The ultrasound probe is inserted through the oesophagus into the stomach and duodenum via an endoscope, similar to gastroscopy. Since the transducer is thus much closer to the structure to be examined and no air overlaps can impair visibility, imaging is improved and even the smallest structures can be detected. In this respect, not only a detailed assessment of the wall layers and structures of the oesophagus, stomach and duodenum is possible, but also a good view of the adjacent organs such as the pancreas.
Endosonography allows a very good imaging of the tissue of the pancreas and the duct structures. Duct dilatations (extensions), calcifications, fluid accumulations, cysts and pseudocysts, necroses, tumours etc. can be easily detected and therefore enable a very precise diagnosis.
Often the additional endosonography can also provide the necessary certainty in the case of a somewhat indistinct finding in CT (computer tomography).
Specific punctures (biopsies) can be performed together with the endosonography, in order to be able to conclusively judge the malignancy of suspicious structures.
Joachim Horcher

New therapy options for pancreatic cysts and small pancreatic tumours
For many years, endosonography has become the preferred diagnostic technique for pancreatic changes. On the one hand, it represents chronic inflammatory changes of the pancreas and allows the diagnosis of benign and malignant cysts and tumours of the pancreas. The fine tissue biopsy of unclear changes is established and firmly anchored in the current guideline of the European oncological society.
In addition to these innovative diagnostic possibilities, endosonography now also offers innovative local therapies. In the last two years, a collection of cases of radiofrequency ablation of both mucinous cysts and small neuroendocrine tumours has built up. Pai et al's group describes radiofrequency ablation of both mucinous cysts and small neuroendocrine tumours with good results and low side effects as early as 2015. Other publications have successfully described radiofrequency ablation of neuroendocrine tumours in small case collections.
In radio frequency ablation, the small tumour centre is punctured by endosonography and a radio frequency probe is inserted into the tumour via the puncture needle. After a slight retraction of the puncture needle, a small amount of current is applied for up to 120 seconds, which leads to local overheating and thus to the destruction of tumour cells. The results were promising and the complications reported so far were minor and corresponded to mild pancreatitis.
"Surgical sanitation"
Another important disease of the pancreas is the lateral duct IPMN, the intraductal papillary mucinous neoplasia. This cystic change, which is associated with the main biliary tract, can lead to classification into high-risk and worrisome cysts based on the so-called Fukuoka criteria. Patients with worrying criteria have a 4% risk of developing pancreatic cancer within five years. In contrast, those patients with high-risk criteria have an almost 50% risk of developing pancreatic cancer within five years. The established therapy for these changes is the surgical treatment of the cysts, the extent of which depends on the size and location of the cysts in the organ.
Nevertheless, we repeatedly see patients who cannot be operated on for various reasons. This can be a very old age, a rejection of surgery or accompanying diseases. Up till now, these patients have not been adequately cared for. A new study published in 2017 by the Moyer et al working group shows the success of cyst ablation through the introduction of a chemotherapeutic agent. The IPMN cyst is punctured by endosonography, emptied via the puncture needle, rinsed with saline solution and then a mixture of the chemotherapeutic agents Paclitaxel and Gemicitabin is introduced directly into the cyst.
In 85% of the cases there was a significant reduction in the volume of the cyst, whereby in 67% of the cases the cyst could no longer be detected in the course of time. The only complication described was acute pancreatitis in the entire group, which corresponds to a risk of less than 5%.
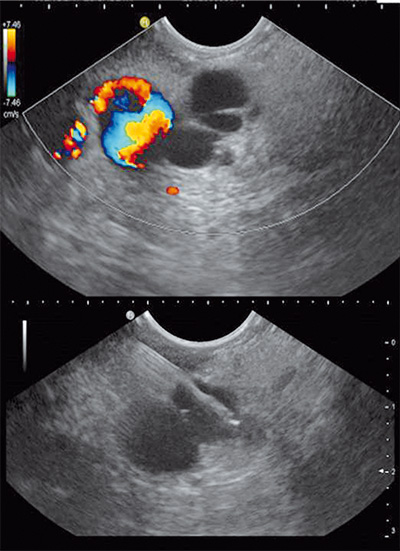
Fig. 2: SD-IPMN - fine needle puncture Image: Prof. Dr. med. Carl C. Schimanski
First experiences succesful and promising
This technique will certainly not be able to replace the operation of cysts in all patients, but offers patients who are not accessible to surgery a good therapeutic alternative. The value of this technology will have to be further investigated in the future.
The experience gained at the Darmstadt Clinic in this regard is encouraging and promising. We are pleased to present this innovative technology.
Prof. Dr. med. Carl C. Schimanski